
**** Pre-Investment ****
There is a sum of €1.56m that is indicated on the status bar that is being invested through third parties in this round.
€650k from a Irish VC
€350k from Enterprise Ireland
€560k from other investment houses or investment syndicates
Business Model
The company is in pre-revenue stage.
It will develop multiple products from its DerMap microneedle patch platform, with the lead product being ITZ-DerMapTM to treat nail fungus. This first product will be commercialised through a partnership with a larger pharmaceutical company active in the dermatology field. The partnership will provide revenue streams for ArrayPatch, including upfront payments, contingent payments and royalties.
Products and Services
ArrayPatch’s initial product from its DerMap platform, ITZ-DerMapTM, is a pain free dissolvable microneedle patch which, upon application, penetrates the outer layer of skin and starts to dissolve and release the medication at the site of infection to kill the fungi. In a process similar to the production of glass from silica/sand, the microneedles completely consist of the meltable drug in the absence of any stabilising material (polymers).
Treatment regime entails weekly microneedle patches that patients apply and wear up to eight hours. Microneedle patches are 1cm in diameter and are comprised of 100% of the drug (generic anti-fungal drug Itraconazole in this case). The patch is applied directly to the site. As a generic drug is being delivered, the development time, risk and costs is significantly lower compared with new drug development.
Problem solved
Nail fungal infection (onychomycosis) that affects both fingernails and toenails is generally chronic and resistant to treatment. Symptoms may include white or yellow nail discoloration, thickening of the nail, and separation of the nail from the nail bed. The infection is usually caused by the same fungus that causes athlete’s-foot. According to the British Association of Dermatologists, 1 in 4 people can be infected with onychomycosis at any given time. This ratio will increase to 1 in 2 for elderly and diabetic patients. It is currently treated with surgery, medical devices (e.g., laser therapy), oral or topical anti-fungal agents or a combination of both. However, patient outcomes are poor – just 1 in 3 cured. Also, treatment times are typically last 12 months with low levels of patient compliance and an associated side-effect of severe liver toxicity.
Based on preclinical proof of concept, ITZ-DerMapTM is expected to deliver superior outcomes – 2 in 3 cured. Shorter treatment times of 3-6 months and better patient compliance with lower toxicity impacts.
Current Traction
- Patent underpinning DerMap delivery technology filed in 2020
- €1.3 million of grant aid funds secured (prior to spin-out)
- ‘Innovation of the Year’ - Pharma Industry Awards 2022
- ‘Commercial Activity of the Year’ - SSPC 2022
- ‘Ireland’s Top 100 Hospital and Healthcare Achievers - HPN Professional 2022
- 1st prize at the "1st Start-up Retreat" Porto, Portugal 2022
- Winner of 2023 IDEATE Ireland competition
- Peer review publications 2023 showing positive research results
- Spun out from School of Pharmacy, University of Cork, in 2024
- Pioneer Group Accelerator programme 2024
- Finalist of Enterprise Ireland Show Case 2024
- Winner of InterTradeIreland Seedcorn Competition 2025
- Interest from word leading microneedle manufacturer to manufacture and suppy lead product
- Positive engagement with European regulatory bodies regarding pending Phase I clinical trial
Partnerships
Research – the company has strong research association with UCC where much of its preclinical proof of concept studies are conducted
Manufacturing - the company will out-source the GMP manufacturing and clinical supply of the lead product to Kindeva USA, a leading global microneedle manufacturer (https://www.kindevadd.com/)
Clinical – the company will secure the services of a leading clinical research organisation (CRO) based in Sweden to support execution of the clinical trials for ITZ-DerMapTM. The CRO has experience conducting human trials involving microneedle technology.
USP of lead product ITZ-DerMapTM
- Better patient results – data suggests 2 out of 3 to show cure response versus only 1 in 3 for existing creams and pills.
- Reduced treatment time of 3-6 months is a significantly faster response time.
- The DerMap patch is just the size of a small coin, applied only once a week and, because of vastly more efficient 100% delivery, it reduces toxicity and promises far higher patient compliance.
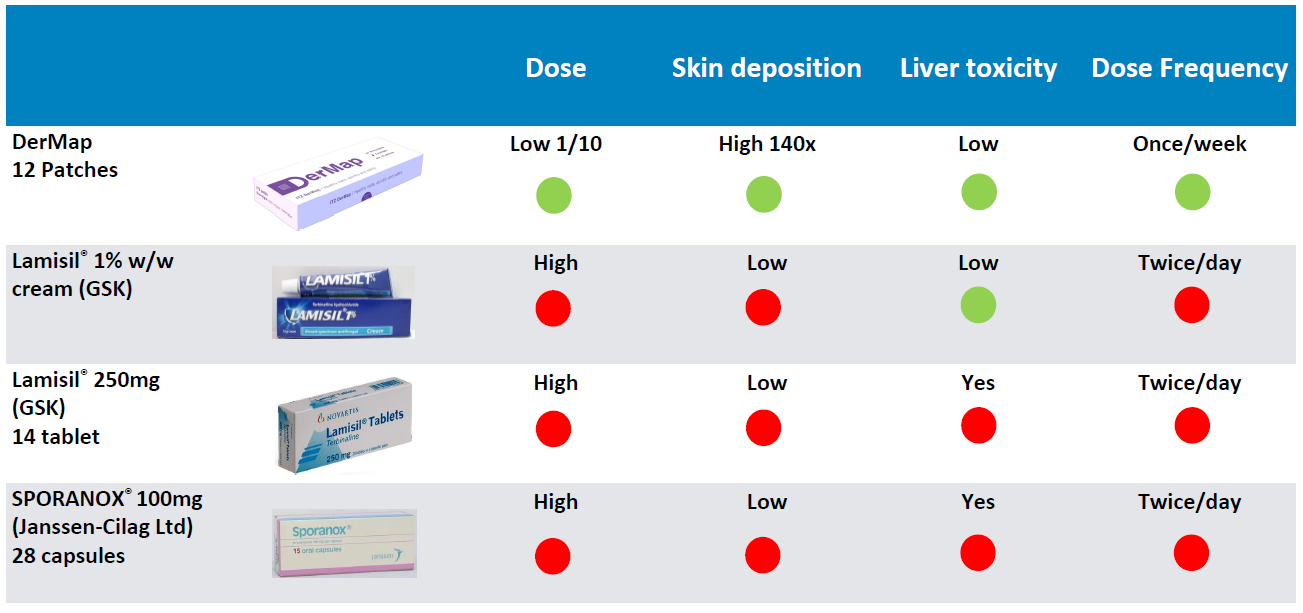
Competitors
Market size to be addressed
- The British association of dermatologists reported that 1 in 4 people can be infected with nail infection at any given time, this ratio increases to 1 in 2 for elderly and diabetic patients.
- Global antifungal drugs market (TAM) was $4.7B in 2022 and estimated to reach $6.2B by 2027, growing at a CAGR of 5.6%.
- North American & European market expected to account for 60%, $3.7B by 2027, of which topical treatments constitute 40%, $1.4B (SAM).
- The company has estimated peak global revenues for ITZ-DerMapTM in the region of $300 million.
Past funding details
€1.3 million from Enterprise Ireland (pre-spin out)
€255K in convertible loan notes (will convert with seed round)
Expansion and growth plans
| Manufacturing | |
| Scale-up production under GMP for human trials | Q3 2026 |
| Clinical | |
| Human usability and acceptability study | Q3 2026 |
| Phase 1 safety/ dosing study – first patient dosed | Q2 2027 |
| Phase 2/3 bridging study (efficacy) – first patient dosed | Q2 2028 |
| Regulatory | |
| European Medicines Agency (EMA) marketing authorisation | Q4 2029 |
| FDA approval | Q4 2029 |
Justification of company valuation
- Founders/management believe, on obtaining positive early P2/3 clinical data, that there is a realistic chance of acquisition or collaboration with an established partner. This could achieve exit returns for investors of more than 10x. Previous examples of acquisitions in the anti-fungal market.
- Pfizer Acquisition of Amplyx Pharmaceuticals - In August 2020. The company was valued at $189 Million. Pfizer framed the acquisition as part of its continuous search for opportunities to build its portfolio of anti-infective/antifungal therapies.
- Basilea Pharmaceutica Collaboration with Pfizer - In September 2017. The deal involved an upfront payment of $70 million, potential milestone payments, and royalties on sales. Under the terms of the agreement, Pfizer will have exclusive rights to develop, distribute and commercialize CRESEMBA, antifungal agent.
- Pfizer's Acquisition of Anacor Pharmaceuticals In June 2016, the deal, Pfizer acquiree Anacor for $99.25 per Anacor share, in cash, for a total transaction value, net of cash, of approximately $5.2 billion, and aimed to strengthen Pfizer's dermatology portfolio, including treatments for onychomycosis.
Planned utilisation of crowd-sourced funds
- Manufacturing scale up
- Funding of preparation, application and conduct of Phase I clinical/human trials with ITZ-DerMapTM.
- Expand management team eg. clinical advisory (dermatology) expertise
